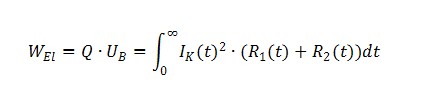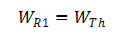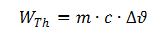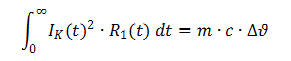Lithium-ion batteries for use in explosion protection
DOI: 10.60048/exm20_03Introduction
The advent of the digital revolution (industrial Internet of Things) has opened up new avenues for reducing costs and increasing system efficiency in the chemical process and petrochemical industries. The need for the comprehensive integration of interconnected digital technology makes the use of suitable portable IT devices such as tablets and smartphones by system personnel essential. Due to the high risk associated with explosive atmospheres, the safety of these mobile devices must be assessed, in particular their batteries.
Secondary batteries (lithium-ion technology) tend to be the energy storage device of choice for portable IT devices. The storable electrical energy, coupled with the material fire load, constitutes a potential ignition hazard in the event that something goes wrong. To enable the explosion protection regulations to describe the potential risks of this technology in greater detail, studies must be undertaken in order to provide a comprehensive assessment of these risks; these studies must look into the various risks associated with the different types of protection. The use of lithium-ion batteries in torches, emergency lighting and industrial trucks must also be discussed [1].
Explosion protection requirements pertaining to cells and batteries as set out in the relevant standards (IEC/EN 60079-0 ff.)
The IEC/EN 60079 series sets out requirements pertaining to equipment destined for hazardous areas, which therefore includes cells and batteries – these differ depending on the type of protection in question. In order to standardise the terminology used, IEC/EN 60079-0, Ed. 7, "Equipment – General requirements", describes cells as a basic functional unit, consisting of an assembly of electrodes, electrolyte, case, terminals and usually separators, that is a source of electrical energy obtained by direct conversion of chemical energy. Batteries, on the other hand, consist of one or more cells and may also, for example, contain monitoring equipment in a special case. Section 23 of IEC/EN 60079-0 has two tables, one for primary (non-rechargeable) and one for secondary (rechargeable) cells, which list the cell chemistries that are permitted for different types of lithium-ion cells. This standard also describes the use of cells or batteries with the same cell chemistries connected exclusively in series. According to the standard, cells or batteries must always be replaced with cells or batteries of the same type, where replacement is provided for. Connecting batteries or cells exclusively in series makes their use in industrial trucks more difficult because industrial trucks require multiple KWh of energy despite a relatively low voltage.
Annex E of IEC/EN 60079-1 defines lithium-ion cells (according to IEC 61960) as used in flameproof enclosures, and describes various requirements such as temperature, monitoring equipment, charging, etc. The cell or battery is accommodated in a case, or enclosure, that is able to withstand the explosion of a combustible gas from within.
Annex G of IEC/EN 60079-2, a standard on protection by pressurised enclosures, describes the use of cells and batteries. The "pressurised enclosure" type of protection prevents explosive atmospheres from forming during operation. The short-circuit test in accordance with IEC/EN 60079-11 is of particular interest here.
IEC/EN 60079-7, a standard on protection by "increased safety", describes the frequent use of devices with cells and batteries. By increasing the dielectric strength and the creepage distances and clearances and reducing the thermal capacity utilisation, we can extend a device's service life and prevent electrical discharge.
IEC/EN 60079-11, a standard on protection by "intrinsic safety", sets out very clearly and in great detail the requirements pertaining to cells and batteries. The standard warns that some types of lithium-ion cells may explode in the event of a short circuit. The standard also describes a short-circuit test with an external short-circuit resistance of just 3 mΩ. In this test, the cell must not be protected by external circuitry. Any external circuitry, which is sometimes found inside an enclosure, must be removed. The cell may have protective mechanisms inside it, but these usually then result in cell failure.
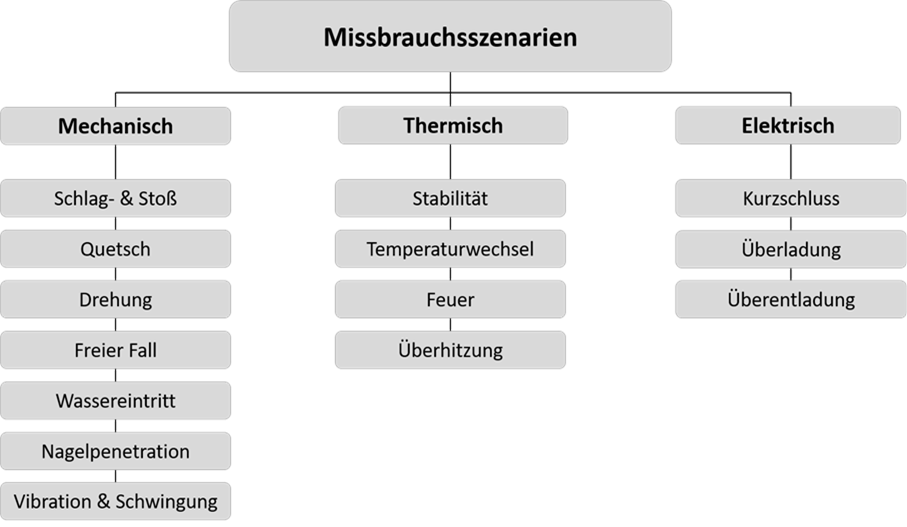
In addition to the explosion protection standards, there are many other standards (e.g. IEC 62133-2 and UL 1642) issued by various standards organisations (DIN, IEC, IEC, UL, SAE, SAND, GB, etc.) that also set out requirements based on use in vehicles, consumer electronics, etc. Due to the importance of vehicles, UN ECE R 100 also provides an overview of the most important requirements, for example the requirements pertaining to the transportation of cells and batteries. UN Recommendation section 38.3 describes in detail a number of relevant tests for rechargeable cells and batteries, which are the basis for the improper use scenarios in Figure 1 below.
A distinction is made between mechanical, thermal and electrical issues. Suitable measures can be used to protect the cells and batteries.
Construction and operating characteristics of lithium-ion cells
Secondary lithium-ion cells consist of a positive electrode, called a cathode, and a negative electrode, known as the anode. Between the cathode and the anode is a substance called the electrolyte, which transports the ions between the electrodes. A separator is used to isolate the electrodes from one another.
During the charging process, a phenomenon known as intercalation takes place, whereby the lithium ions released from the cathode material become embedded in the anode's graphite layer. During the discharging process, the lithium ions are released again and move through the electrolyte back to the cathode. The electrons that are released in this process flow through the consumer via the electrodes' collectors. Materials such as lithium nickel cobalt aluminium oxide (NCA), lithium iron phosphate (LFP), lithium cobalt oxide (LCO) or lithium nickel manganese cobalt oxide (NMC) are used for the cathode. When used in conjunction with a graphite anode, the mean open-circuit voltage is approximately 3.6 V [2]. Strictly speaking, the operating principle does not exactly match the definition of a cell given in IEC/EN 60079-0, which describes a chemical accumulator.
A cell's key parameters include its capacity and its electrical resistance. A cell's capacity is defined as the storable amount of charge in Ah within a specified voltage range. A cell's state of charge (SOC) is stated as a percentage. A cell's state of health (SOH) indicates its loss of capacity over the course of its use. Modern lithium-ion cells allow over 1000 charging cycles before the SOH falls below 80%. At the end of a cell's service life, its usable capacity drops off rapidly.
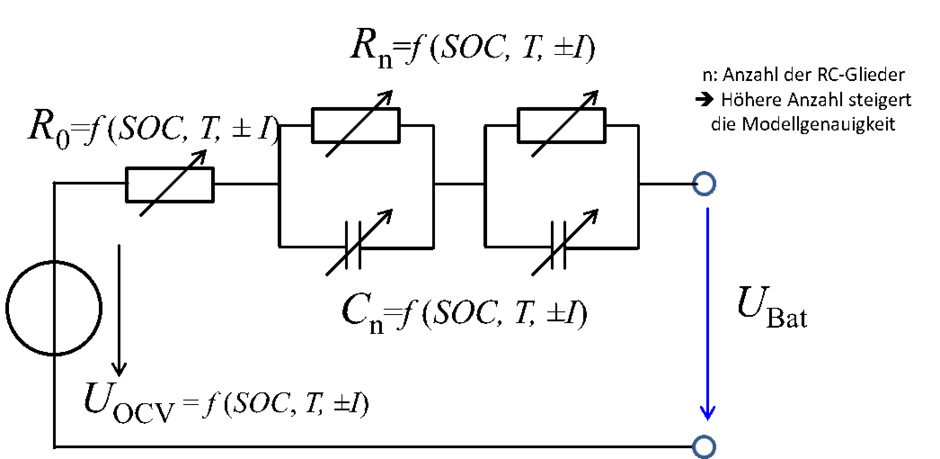
To understand lithium-ion cells, you also need to know about the internal resistance. A simple equivalent circuit diagram (Fig. 2) illustrates a cell comprising a resistor R0 and two RC elements connected in series. [3]
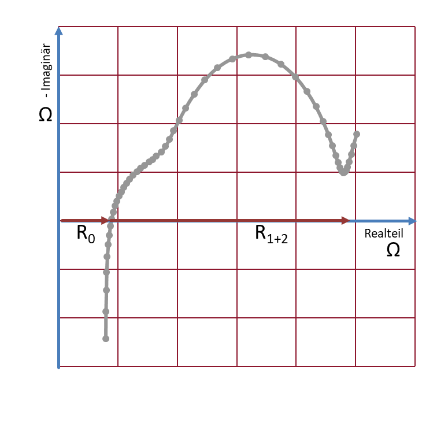
Impedance spectroscopy can be used to chart the behaviour of the alternating current internal resistance (impedance) as a function of the frequency (Fig. 3). The curve on the graph represents the processes within the cell as a function of time, and allows the cell's state of health to be ascertained by comparing its original state to its current state.
Risk associated with lithium-ion cells
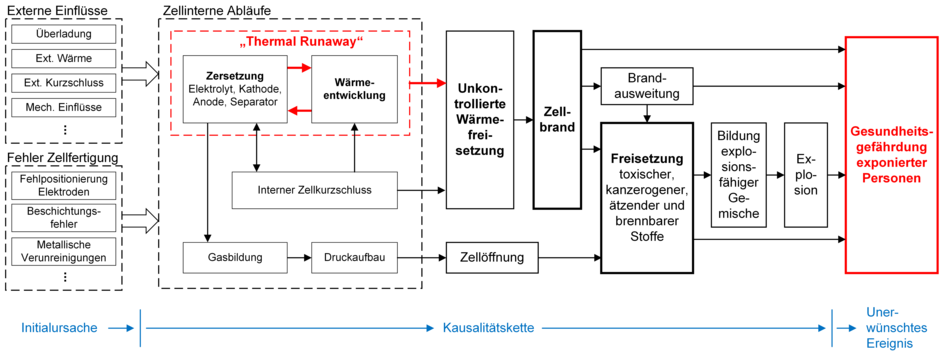
The use of lithium-ion cells in consumer electronics and transportation applications is incredibly widespread, and they are produced in enormous quantities and in a huge range of variants. Unwanted incidents involving lithium-ion cells can occur and have a variety of causes. Temperature is a significant cause of thermal runaway and can instigate a causal chain at the end of which there is a risk to personal safety (Fig. 4).
Possible scenarios include the following, for example:
- Exothermic reactions take place at the anode; these reactions can proceed without further input when the cell's temperature is elevated. They include the decomposition of the SEI (solid electrolyte interface) with the binder, the reaction of the intercalated lithium and the electrolyte and between the binder and the decomposition products from the other decomposition reactions.
- The cathode material is delithiated during the charging process, which reduces its stability.
- NMC (lithium nickel manganese cobalt oxide) with a high nickel content is very vulnerable to overcharging, for example. Even when overcharged just a little, there is a considerable reduction in capacity. Significant overcharging causes the crystal to collapse. When the active material decomposes, oxygen is released, which can result in significant exothermic oxidation and the cell may even catch fire. Due to the oxygen being released by the cell itself, this kind of fire is difficult to extinguish.
For the purpose of explosion protection, devices are assessed on the basis of the zones in which they are to be used. For Zone 2, the device is deemed "safe" if no potential source of ignition exists under normal operating conditions. For Zone 1, this also extends to the existence of moderately likely failures, and for Zone 0, this even includes rare failures. Figure 5 depicts, as examples, a number of potential failure scenarios and the possible categories into which they might fall. A specially tailored approach to categorising the possible failure conditions should be specified in the explosion protection standards. This depends on the equipment used and its application range [1].
By way of example, the categorisation of failure scenarios in terms of the likelihood that they will occur could be implemented as described below. Critical states resulting from over- and deep discharging could be considered to be rare failures. The battery management system (BMS) monitors the charging and discharging processes and prevents states like these from occurring. If the BMS fails, this can be detected by means of self-diagnostics, and the necessary safety steps (e.g. shutting down the equipment) can be taken.
Nevertheless, the battery should not be charged in the explosive atmosphere. Manufacturing faults with long-term implications could also be categorised as rare failures. Stringent quality control by manufacturers could minimise these faults. IEC 62133-2 sets out various requirements pertaining to lithium-ion cells, which should also be applied to explosion-protected devices.
External mechanical influences such as dropping the device onto a hard surface could be considered to be moderately likely failures. Portable IT devices being placed onto hot surfaces and external short circuits caused by the electronics are two further examples of moderately likely failure scenarios. Internal short circuits due to the cell's state of health are another possibility. Initial reactions that produce gas within the cell cause the cell to overexpand.
Under normal operating conditions, the lithium-ion cell heats up, for example by 20 K. This is an important consideration when it comes to deciding the temperature class with respect to the ambient temperature range. Due to their low boiling point and highly combustible nature, a degree of risk is associated with the components of the electrolyte. The thermal decomposition of the conducting salt and the solvents also poses a safety risk.
In terms of basic requirements for use in hazardous areas, the following points should also be taken into account:
- Batteries are subject to continuous voltage and cannot be de-energised. For this reason, enclosures cannot simply be opened in hazardous areas.
- In the event of thermal runaway, the cell produces gases, which will increase the pressure in a sealed enclosure. Initial tests show that 2 dm³ per Ah is to be expected. In a test conducted with a flameproof enclosure, this resulted in the enclosure rupturing.
- In cells with a low internal resistance, breaking sparks that can cause a fire may occur even at low voltages (if the device is accidentally dropped on the floor and the battery falls out).
- The risk of ignition due to electrostatic discharge during normal use of a portable device must be eliminated.
Ascertaining the maximum temperature in the event of a short circuit (test and calculation)
Ascertaining the maximum temperature in the event of a short circuit (test and calculation):
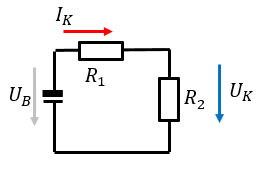
One of the requirements set out in the explosion standards is to ascertain the temperature of the cell in the event of an external short circuit. Whilst many standards specify an external short-circuit resistance of 80 mΩ, EN/IEC 60079-11 specifies a value of 3 mΩ. Because the short-circuit test is conducted on a circuit comprising the cell's internal resistor (R1) and the external short-circuit resistor (R2) connected in series (Fig. 6), the cell's energy is only converted in the cell if R1 » R2.

This calculation can be used as an initial estimate of the overtemperature that is to be expected, but the standard stipulates that a test must still be conducted.
Example short-circuit test
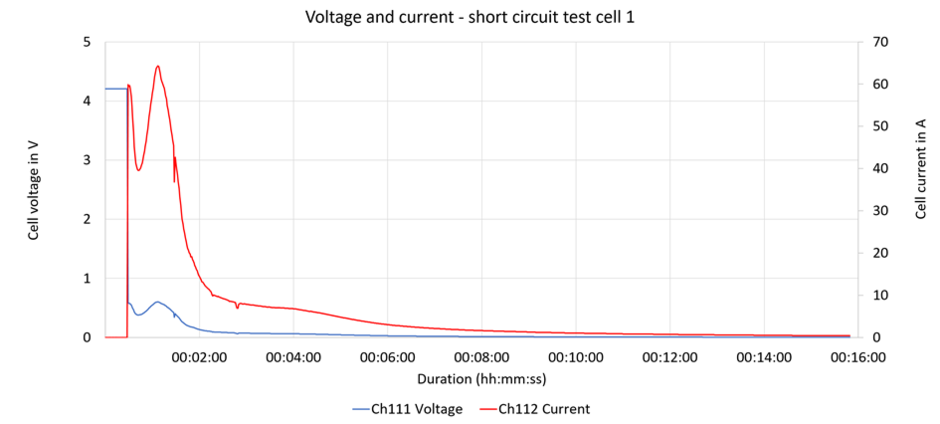
Because the possibility of the cell undergoing thermal runaway during a short-circuit test cannot be completely eliminated, the test should be conducted in an encapsulated enclosure and the gases that are produced should be filtered and conducted away. Figure 7 shows a lithium-ion cell equipped with thermocouples in a flameproof enclosure. It also has battery connection lines and a means of supplying and discharging gas.
The voltage drops significantly when the short circuit occurs (< 1 V), which may indicate that R1 » R2. Within the first 2 minutes, the current increases to up to 60 A which, for a cell capacity of 4.35 Ah, corresponds to approximately 14 times (14C) the nominal current (1C) (Fig. 8). In these first 2 minutes, a charge of approximately 2 Ah is therefore converted to thermal energy (see current and voltage behaviour during short-circuit test in the figure below).
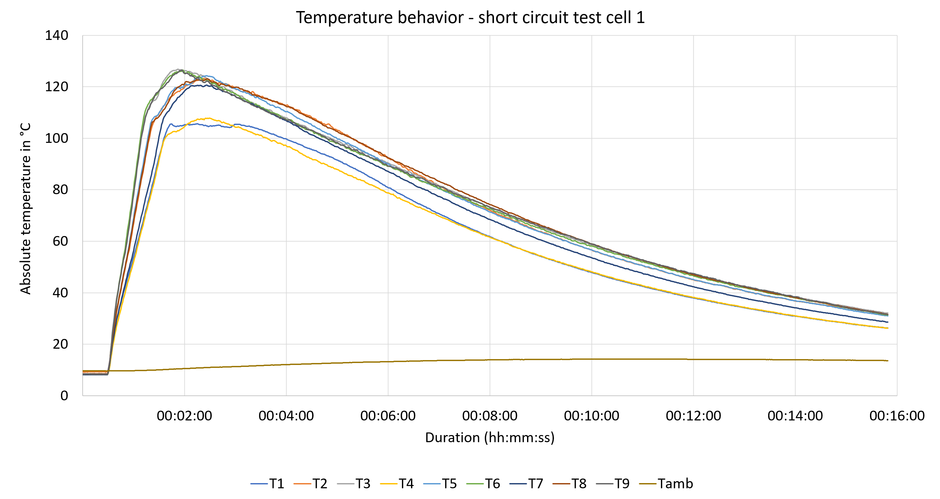
Within 2 minutes, the temperature of the cell's outer jacket rises by approximately 120 K before gradually tapering off (Fig. 9).
The cell has not undergone thermal runaway but has overexpanded due to internal gas production.
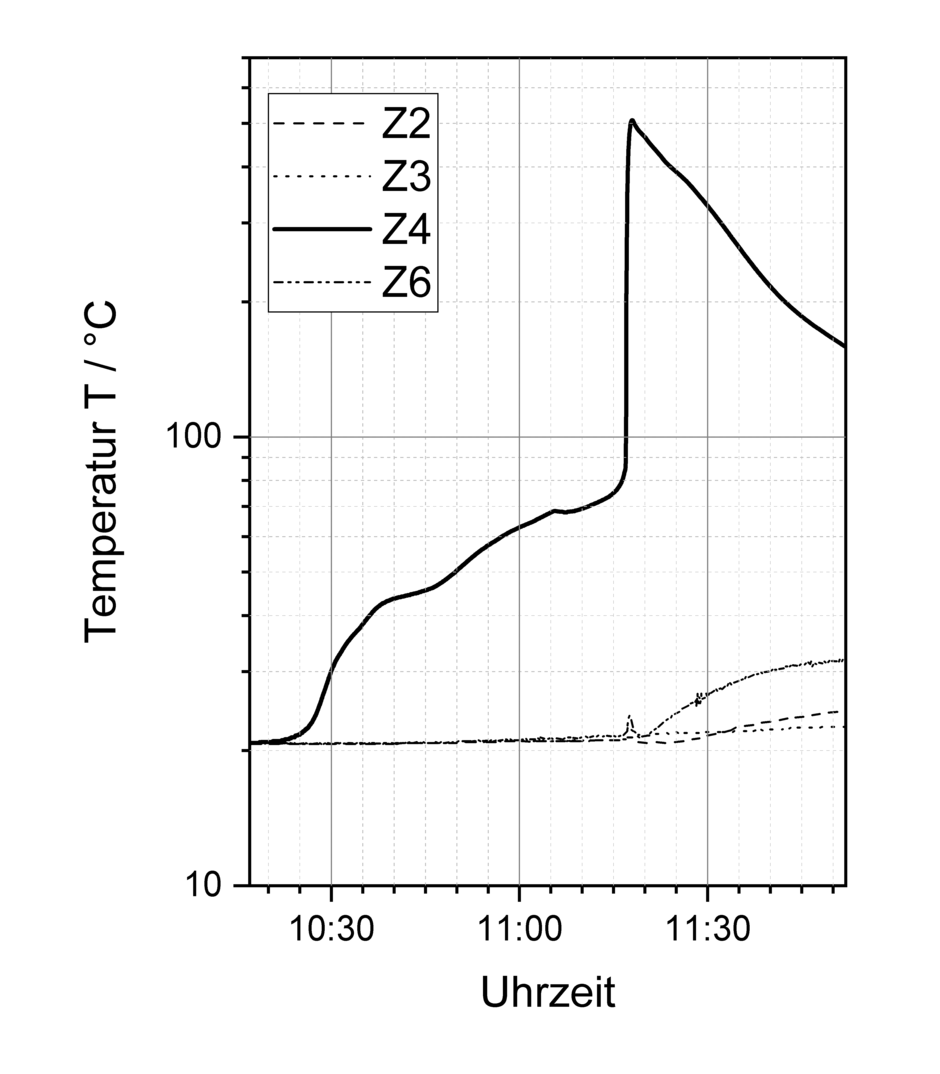
Example overcharging: Overcharging a cell brings about a further critical state. Figure 10 shows the temperature behaviour in a cell assembly. When the cell 4 (Z4) starts to be overcharged, the temperature starts rising continuously and, from a temperature of approximately 80 °C, the cell starts to undergo thermal runaway, resulting in the temperature suddenly jumping to 500 °C. This resulted in the cell rupturing, allowing gas to escape and causing deflagration [6].
Example overcharging
Overcharging a cell brings about a further critical state. Figure 10 shows the temperature behaviour in a cell assembly. When the cell 4 (Z4) starts to be overcharged, the temperature starts rising continuously and, from a temperature of approximately 80 °C, the cell starts to undergo thermal runaway, resulting in the temperature suddenly jumping to 500 °C. This resulted in the cell rupturing, allowing gas to escape and causing deflagration [6].
Safe operation and monitoring of batteries and cells
To ensure that lithium-ion batteries operate safely, the operating condition must be monitored in order to minimise the substantial risk of incidents. This requirement is not explicitly set out in the explosion protection standards. To prevent them from being over- or undercharged, lithium-ion cells can only be operated within a defined voltage range. Due to the low internal resistance of lithium-ion cells, a very high current is able to flow; this current can be many times greater than the operating current. A cell's service life/SOH is affected by factors such as temperature and overload.
Figure 11 shows the operating range of a lithium-ion battery by way of example [4].
The green area is the usage range for extending a cell's service life (SOH), while the yellow area is the cell's operating window. The temperature refers to both the ambient temperature and the self-heating of the cell during operation; given a maximum temperature of 60 °C, the maximum ambient temperature should be significantly below this. Additionally, the cell cannot be operated unless the initial chemical processes that destroy the cell have begun.
The battery management system (BMS) is responsible for monitoring the relevant parameters to ensure that the cell is in a safe operating condition. These parameters include the cell's voltage and current values and its temperature behaviour. The BMS is an integral part of all mobile phones and tablets. Its fitness as a safety device for ensuring that the explosion-protected device operates safely throughout its service life must be assessed.
Summary
The use of lithium-ion cells is incredibly widespread, and they are increasingly being used in hazardous areas. In order to meet the stricter requirements of explosion protection applications, an assessment that incorporates not only normal operation (Zone 2) but also moderately likely failures (Zone 1) must be carried out. The requirements of current standards are still very substantially geared towards conventional cell types, which means that there is still work to be done when it comes to special types of lithium-ion cells.
References
[1] M. Losch; A. Stein; T. Horn; F. Lienesch: Explosionsschutz als Herausforderung für portable IT-Endgeräte [Explosion protection as a challenge for portable IT devices]. 15th BAM-PTB symposium. DOI: 10.7795/210.20190521Q. Brunswick 2019.
[2] R. Korthauer (ed.); S. Leuthner; K. Vuoritehto; M. Fleischhammer; H. Döring; H. Sattler: Handbuch Lithium-Ionen-Batterien [A guide to lithium-ion batteries]. Springer-Verlag Berlin 2013.
[3] U. Westerhoff, K. Kurbach, F. Lienesch, M. Kurrat: "Analysis of Lithium-Ion Battery Models Based on Electrochemical Impedance Spectroscopy", Energy Technol., vol. 4, no. 12, pp. 1620-1630, 2016.
[4] Kokam: SLPB (Superior Lithium Polymer Battery) Technical Specification. May 2010.
More Article
Ex in sight
![[Translate to Englisch:] [Translate to Englisch:]](/fileadmin/user_upload/magazin/artikel/20_27_Ex_im_Blick/1_Ex_im_Blick_Teaser.jpg)
Incorrect storage of hazardous chemical substances and inadequate surveillance of storage areas all too often result in catastrophic…
The Need for Integrated Project Management

Reducing project management and development time alone has the potential to deliver 15 to 30 percent in cost savings
Static and dynamic material stresses acting on Ex "d" enclosures

Flameproof enclosures must be subjected to certain testing, including of their ability to withstand pressure
PLP NZ celebrating 45 years with R. STAHL

45 years ago, three things came together, R. STAHL, PLP (Electropar Ltd), and the willingness to adopt innovative new hazardous area…
Emergency lighting

Central battery systems as emergency lighting systems offer secure protection in the event of a power supply failure
Sensing nonsense: When appearances are deceptive

Process engineering systems are generally controlled by measuring process variables such as temperature, pressure, quantity, fill level or…
Digital support for visual inspections using deep learning

The use of deep learning models offers huge potential for reducing the error rate in visual inspections. Smart object recognition enables…
Lightning and surge protection in intrinsically safe measuring…

According to Directive 1999/92/EC [1], the user or employer are obliged to assess the explosion hazard posed by their system and they must…
Ex assemblies, Part 1

The discussion about Ex assemblies is as old as the EU ATEX Directive, and now dates back almost 20 years
How R. STAHL TRANBERG is Meeting the Digitalization Demands of…

Digitalization and the integration of data and solutions is playing a pivotal role in the shipping and maritime industries today, having a…
The "PTB Ex proficiency testing scheme"

The "PTB Ex proficiency testing scheme" (PTB Ex PTS) is a project that involves developing interlaboratory comparison programmes to assess…
Non-electrical explosion protection

Manufacturers and users must acquire knowledge on the subject of non-electrical explosion protection, in order to assess the application,…
Certification in South Africa

Certification in South Africa has certain key differences from international certification, e.g. IECEx or ATEX
Global conformity assessment using the IECEx system
![[Translate to Englisch:] [Translate to Englisch:]](/fileadmin/_processed_/c/e/csm_Einstiegsbild_IECEx_Paragraf_38414b50d0.jpg)
IEC Technical Committee (TC) 31, tasked with developing a global conformity assessment system for explosion-protected products
A mine of experience in industry
![[Translate to Englisch:] [Translate to Englisch:]](/fileadmin/_processed_/f/f/csm_Einstiegsbild_e_tech_IEC_6270d7c66a.jpg)
He takes over from Thorsten Arnhold, who chaired the System for the past six years
Conformity assessment in the USA

In contrast to the international IEC/IECEx community and the European Union, the conformity assessment landscape in the USA is very…
25 Years of the Zone System in the USA

In the area of explosion protection, the publication of Article 505 in the 1996 National Electrical Code (NEC®) was seen as a giant step…
An Ex-citing future with hydrogen
![[Translate to Englisch:] [Translate to Englisch:]](/fileadmin/_processed_/0/1/csm_shutterstock_1644506059_7d60f29da8.jpg)
Aside from a few exceptions based on the effect of gravitation and radioactivity, hydrogen is the source of most primary energy that makes…
Certification of Ex products
Since 1st October 2019, new CCC certification rules have been in place for Ex products sold in China
LNG - The key energy source in the medium-term on the way to…

LNG stands for Liquefied Natural Gas and refers to natural gas that liquefies as it deep-cools to -162 °C and thereby shrinks to 1/600 of…